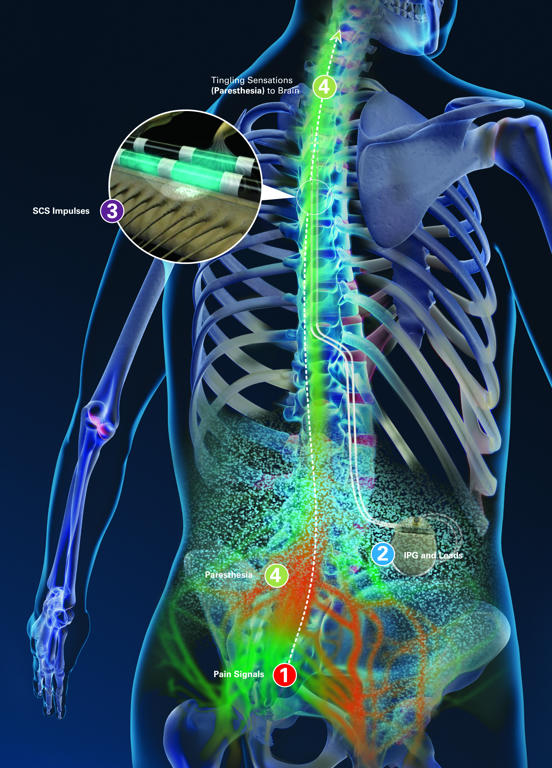 
Do not perform an MRI scan on patients who have an incomplete neurostimulation system, where a lead is present without the IPG or disconnected from the system. Serious patient injury could occur.Ībandoned devices. Do not perform an MRI scan on patients who have any components of a neurostimulation system that are unapproved for use in an MR environment. The following warnings, precautions, and potential adverse events apply when performing MRI scans on Abbott MR Conditional SCS/DRG systems. IPG: Upper buttock, low back, flank, or abdomen IPG: upper buttock, low back, midline, flank or abdomen IPG: upper buttock, low back, midback, flank or abdomen MRI Scans with Spinal Cord Stimulation or Dorsal Root Ganglion Stimulation.See product advisories.įor expanded patient instructions on how to prepare for an MRI scan, please see the links below: Note, the Class I recall on the Proclaim™ Family of devices and the Infinity™ DBS System initiated in July 2023 does not impact a patient's eligibility for receiving an MRI scan, the conditions for the MRI eligibility, or patient safety in an MRI environment. That's why we’ve designed our devices and technologies to fit as seamlessly as possible into this critical step in your diagnostic workflow. Certain Abbott neurostimulation systems are MR Conditional with 1.5T closed bore scanners subject to conditions in the MRI Procedures Manual.īefore your patient undergoes an MRI scan:Ĭonfirm the MR Conditional components and location of the system (this information is included on the Abbott Patient ID card).Ĭonfirm that no adverse conditions to MR scanning are present.Įnsure the patient’s neurostimulation system is in MRI mode.Ĭonfirm specific scan requirements for the patient's system, intended scan location, and RF coil type. Stimulators are also being used on patients with Complex Regional Pain Syndrome (CRPS).We know that MRI images are used as a tool to visualize diagnostic imaging and help provide information that may be useful to a clinician. Last year the FDA expanded the use of stimulators to include lower limb pain from diabetic neuropathy. SCS’s are no longer limited to patients with intractable back and neck pain. The review also identified nearly 500 deaths linked to the devices, along with nearly 78,000 injuries and 30,000 malfunctions. An FDA review of adverse events involving stimulators found that nearly a third were reports of poor pain relief. 
About a fifth of the patients experienced complications so severe the devices had to be removed or revised.Ī 2018 study found that SCSs have some of the worst safety records of medical devices tracked by the FDA. Researchers at the University of California San Francisco School of Medicine found that pain patients with stimulators did not reduce their use of opioids, epidurals, corticosteroid injections or radiofrequency ablation compared to a control group. and their use is growing – in part because of the belief they’ll reduce the need for opioids and other pain therapies.Ī recent study found that premise to be untrue. The devices can also been monitored and programmed with a mobile app.Ībout 50,000 SCSs are implanted annually in the U.S. This makes a big difference in comfort for many patients who now can have access to the best of both worlds – a small, best-in-class rechargeable device with superior stimulation therapy."Īnother advantage of the Eterna SCS system is that patients with the device can still have full-body MRI scans – a critical need for those who need access advanced imaging and diagnostics. "Until now, it wasn't available on a rechargeable device that was this small, and that only needs to be charged a few times a year.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
